 Potassium hydroxide (KOH), typically about 6M, is the most used alkaline electrolyte for ZABs because of its high conductivity and activity for both Zn and air electrodes. 2(a), a typical Zn–air battery (ZAB) is composed of four main components: a Zn anode (Zn metal, Zn paste, or Zn powder), an air-breathing cathode, an alkaline electrolyte, and a separator. 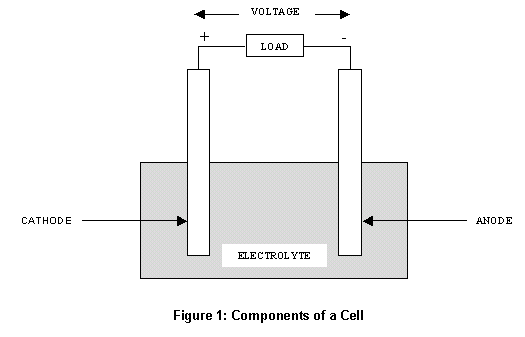
7,8 ZABs acquire a unique half-open system with cathode utilizing oxygen from ambient air as the active material, which decreases the total mass and the volume of the cathode. 5,6 Additionally, Zn metal is earth-abundant and highly resistant to corrosion in alkaline solutions, giving ZABs outstanding advantages such as low capital cost, safe fabrication process, flat discharge voltage, and long shelf-life. 1–4 Despite the slow progress, ZABs have always been considered as a highly promising battery system for electric vehicles and portable applications, since their theoretical gravimetric and volumetric energy densities ( 500 W h kg cell − W h l cell − 1) are higher than those of the commercialized lithium-ion battery (LIB) (350 W h kg cell − 1 and 810 W h l cell − 1), as shown in Fig. The final part summarizes the properties of air electrodes needed to fulfill the requirements of electrically rechargeable ZABs and provides ideas for the future designs of air electrodes.Īlthough intensive research on electrically rechargeable Zn–air batteries (ZABs) began in early 1960’s, a commercial breakthrough has not been accomplished yet due to the inferior overall energy efficiency and poor cyclability, which is correlated with the difficulties present in optimizing the cathode and anode components. In-depth discussion of O 2 catalysts for air electrodes and progress of binder-free air electrodes for ZABs are presented in the following based on three major modification strategies: defect engineering, cation/anion regulation in multi-components transition metal compounds, and single or multi-heteroatom doping in carbon materials (metal-free and metal-based material). The first part describes the fundamentals of an ideal air electrode with its corresponding oxygen electrochemical reactions and typical bifunctional O 2 catalysts. This review highlights the structural features essential to satisfy the design of the cathode compartment of ZABs and presents the associated factors that drive the oxygen reactions in the air electrode based on the relationship between the intrinsic activities of bifunctional O 2 catalysts and the collective strategies employed to modify the electronic structure of such electrocatalysts. To develop a structural design that could provide accessible active sites to oxygen, electrolyte, and electron, it is necessary to modify the overall structure of an air electrode, which is considered as the most significant and complicated part of Zn–air batteries (ZABs). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
